 In addition, adsorption refrigeration facilitates the use of renewable energy sources, for example, solar, wind, or tidal power, (3,5) which are expected to increase in the near future. The adsorptive process enables us to replace mechanical compressors in vapor–compression cycles, reducing energy consumption. (3,4) In adsorption refrigeration systems, the cooling effect is driven by the thermal energy produced and injected into an adsorbent bed. Adsorption refrigeration has been proven to be an excellent potential candidate for replacing conventional, energy-intensive vapor compression refrigeration cycles. (2) Therefore, efficient and environmentally friendly refrigeration cycles are of utmost importance. Therefore, this study guides the selection of MOFs suitable for thermal-storage applications with this new class of low-GWP refrigerants, helping in meeting cooling global demand combined with intermittent sources of energy in a step toward achieving the sustainable energy scenario.ĭue to global warming and boost of wealth in tropical regions, the demand for refrigeration and air-conditioning is likely to increase in the upcoming years, (1) a process that already accounts for around 17% of the global electricity consumption. It is also observed that the M-MOF-74 family is not suitable for CTES under the given operating conditions, but some of them may be appropriate for TES applications. 
Conversely, MOFs presenting large pore sizes, such as Cr-MIL-101 and IRMOF-10, MOF-200, have a low affinity for HFO and large working capacities, showing a considerably higher CTES energy density than the currently used activated carbons/R134a pairs. Results show that MOFs with open metal sites have a strong interaction with R1234yf and R1234ze(E), making them more suitable for TES. We conducted Grand Canonical Monte Carlo simulations to establish a relationship between the adsorptive capacity and material properties. A total of 40 MOFs, belonging to several representative structural families were studied, including IRMOF, M-MOF-74, ZIF, COF, NU, and MIL topologies. The choice of these refrigerants is based on the need to deploy low-GWP refrigerants after the ratification of Kigali’s agreement, suitable for different cooling applications. For comparison, the third-generation refrigerant R134a currently in use is also considered. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
In this work, we have conducted the first computational screening of experimentally available metal–organic frameworks (MOFs) for CTES and TES units using three low-global warming potential (GWP), fourth-generation refrigerants: hydrofluoroolefin (HFO) R1234yf, R1234ze(E), and the blend R513A, in order to search for the best MOF-refrigerant pair for this application. In CTES and TES applications, the choice of a suitable adsorbent-refrigerant working pair plays a crucial role. Such adsorption-based refrigeration cycles facilitate the application of renewable energies and allow energy storage, that is, both cold thermal energy storage (CTES) and TES. 6.Refrigeration processes based on physical adsorption are promising candidates for replacing high energy-intensive vapor compression cycles.6.2 Classifying the Elements - 6.2 Lesson Check.6.2 Classifying the Elements - Sample Problem 6.1.6.1 Organizing the Elements - 6.1 Lesson Check.6.1 Organizing the Elements - Chemistry & You.6.1 Organizing the Elements - Chemistry & You: Everyday Matter - Take It Further.Next Answer Chapter 6 - The Periodic Table - 6.3 Periodic Trends - 6.3 Lesson Check - : 24 Previous Answer Chapter 6 - The Periodic Table - 6.3 Periodic Trends - 6.3 Lesson Check - : 22 Will review the submission and either publish your submission or provide feedback. You can help us out by revising, improving and updatingĪfter you claim an answer you’ll have 24 hours to send in a draft. 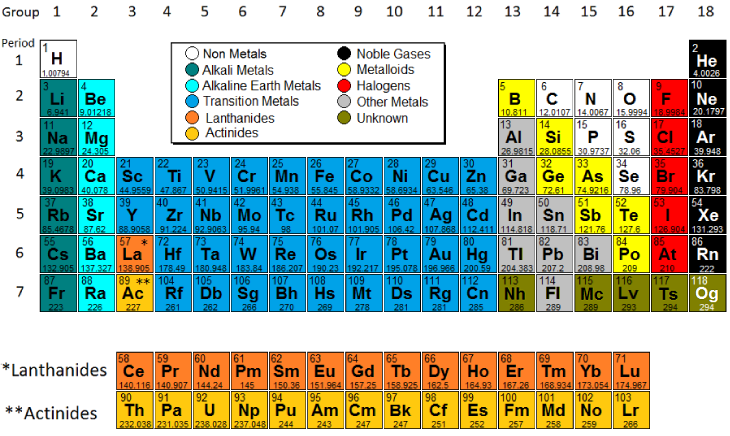
These variations in structure are what give atoms many of their properties, and the nature of the periodic table provides for periodic trends that illustrate atomic properties. Lastly, ionic size depends on the different orbitals and energy levels of atoms. 
The opposite idea, that of atoms pulling electrons from another atoms (electronegativity) can be explained similarly. Ionization energy depends on similar ideas, because the stronger the attraction between protons and electrons, the harder it is to get an electron away from an atom. For example, atomic size depends both on the number of protons (and how strongly these protons attract the cloud of electrons) and the number of energy levels occupied by electrons. Periodic trends displayed by the elements can be explained by structural variations of the atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
